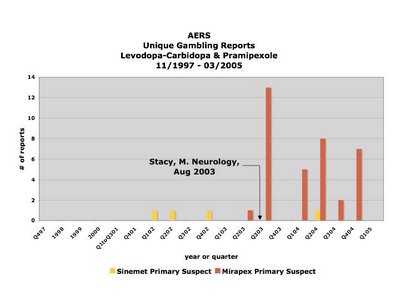
The above is a graph of the distribution over time of the registering of reports linking gambling to either levodopa-carbidopa/Sinemet (yellow) or pramipexole/Mirapex (red) to the FDA's Adverse [Drug] Event Reporting System (AERS) database between November of 1997 and March 2005. The publicizing of the first study purporting to have found an association between pramipexole/Mirapex is denoted in August, 2003, and its effect is clear.
And here is the context for that bar chart.
Ana Szarfman, MD, PhD, P. Murali Doraiswamy, MD, Joseph M. Tonning, MD, MPH, and Jonathan G. Levine, PhD published a research letter to the editor entitled “Association Between Pathologic Gambling and Parkinsonian Therapy as Detected in the Food and Drug Administration Adverse Event Database” in the Archives of Neurology in February of 2006.
In their letter, Szarfman et al say they found, among other things, 39 reports linking Mirapex to gambling in the FDA's AERS database. They then went on to apply the Multi-item Gamma Poisson Shrinker (MGPS) statistical algorithm to the reporting rates of certain drugs in conjunction with gambling. I am not sure how that works, but Mirapex came out the other end of that algorithm looking guiltier than ever.
However, what Szarfman et al failed to reveal was that all but one of the reports in question regarding Mirapex were registered after the first study purporting an association between it and gambling was publicized heavily by the AAN in August of 2003 (Stacy, Driver-Dunckley. Neurology, August 2003).
The fact that Szarfman et al chose not only not to reveal but also not to take what is known as "the publicity effect" into account, particularly one of such magnitude (there was a virtually 100-fold increase in the reporting rate starting immediately after the publicizing of the study in 2003, and this was after extensive clinical trials and six years on the market with only one gambling report) is at odds with a recommendation she and her co-authors make in their 2005 paper entitled “Perspectives on the Use of Data Mining in Pharmacovigilance,” in which they say:
"Publicity resulting from advertising, litigation or regulatory actions (e.g. "Dear healthcare provider" letters and product withdrawals) may result in increased reporting and can generate higher-than-expected relative RRs.[reporting ratios][56,58] Relative RRs should be examined over time in hopes of detecting these influences, although there are no definitive criteria for using data-mining techniques to reliably identify such effects."In this case, the number of reports is so small number and they were registered over such a short period of time that there was no need to use data-mining techniques to detect this effect. In fact, it would be impossible not to see such a sudden skyrocketing in reporting as this, if one were in possession of the data - as I am, having requested it directly from the FDA when Dr. Szarfman declined to provide me with the dates the reports were registered.
Consequently, is difficult to interpret it as anything but disingenuous when Szarfman et al state in their conclusion that the reports stimulated by the reporting of the case series from the 2003 study to the AERS may have increased the MGPS signal scores. It would be difficult to argue against the statement that the stimulated reporting was the MGPS score, and that in fact without it, there would be no MGPS score for Mirapex at all.
If anyone wants to go through the raw data, please contact me – I would be very happy to send it to you – or my refined data – yours for the asking. The dates of the reports don’t lie.
4 comments:
I have so much to say on this, but typing is very difficult sometimes. Needless to say, Mirapex (an Agonist), was very good at addressing my specific symptoms associated with PD. However, over the period from late 1997 through June, 2005 when I stopped taking Mirapex, I was ruined........obsessive compulsive behavior of several types, most disastrous of which was the gambling.. I lost my marriage, lost a 21 year carreer a Chevron Corporation, lost all of my savings, 401K, ruined my credit, went through bankruptcy, and have suffered most recently with having no home of my own for the last 13 months, I struggle to work (must commute 105 miles to work (1 way) each week) because my reputation is ruined, and have to rely on county medical services, because I cannot afford private medical insurance.
........this all because no notice or mention was made that this particular agonist could cause side affects of a serious nature........obsessive compulsive behavior..........who would have thought that a prescribed medication could lead a person to financial ruin?
I have much to say, as well - it will all be in my blog at some point - will reply in greater detail tonight.
I don't think what i have to say will go over well with you, but please be assured that whatever i say, it does not indicate lack of sympathy with your experience, which was and is totally devastating, obviously. However, the reason no mention was made of any association between compulsive gambling and mirapex is because there was none - not even a hint of one - in fact, only one report associating the two came into the FDA's adverse event reporting system (AERS) database prior to the publication of the first gambling study in 2003 (which actually failed to find an association, if you do the math), and that came in only a month before the study was published. that means there was zero indication in clinical trials, and for the first six years Mirapex was on the market, zero reports to the AERS. During that same time period, there were - can't remember how many exactly but somewhere between 2 and 5 reports associating gambling with levodopa.
The alleged "FDA" study failed to reveal that there would have been no "score" for Mirapex whatsoever had it not been for publicity-generated reports (generated by a study that did not find an association, despite all of the publicity to the contrary) and certainly did not take that into account when intperpreting the reporting rate.
But i am getting ahead of myself. Bottom line is this - I am not saying what happened ot you wasn't real, and i am not saying it wasn't and isn't devastating - it *could* even have been the fault of Mirapex. What I am saying is that none the three major studies that have come out since 2003 actually found an association between the two.
Hi,
I know of others who believe their gambling has increased due to agonists. With all due respect to all of them, I find it difficult to accept that it has been proven.
As this blogger points out, putting Mirapex out of business seems more dangerous to the pwp community than inconclusive studies claiming its' responsibility for the development of gambling habits.
Obsessive compulsive behavior is as individual as any other aspect of Parkinson's Disease. Will inidviduals begin blaming it for other compulsions - such as theft? At what point does personal responsibility end?
Again, I mean no disrespect to Wayne. But I do have a question about these studies. Did they identify where or how the gambling was done? Specifically, was it on the Internet?
Anuket,are you still there? I don't see any recent posts, and I want to know more about all of this
Bob Dawson
dawson.schulz@gmail.com
Post a Comment