If you have read any of my recent posts, you will know that I don’t think too highly of the drug most commonly prescribed for Parkinson’s, levodopa/carbidopa (often referred to as l-dopa). Because of its obvious deficits, I have marveled at its having acquired the moniker “the gold standard,” and have long since concluded that it must have been a stroke of genius on the part of some marketing person, probably at a time when the drug’s public image needed a boost. This weekend, I decided to try and figure out who first used the term, and when. The following is what I found.
The term "the gold standard" with reference to levodopa makes its first appearance in PubMed in 1996, in a study by a fellow named Mark D Lindner, sponsored by company called Cytotherapeutics:
" Although the therapeutic gold standard, L-DOPA:carbidopa (Sinemet), effectively attenuates parkinsonian symptoms..."(1)
And then, again, in 1997, the second time “the gold standard” appears on PubMed, Mark D. Lindner is one of the authors:“
Guidelines for clinical transplantation studies for Parkinson's disease emphasize that transplants should be considered as an adjunct to systemic L-DOPA, yet few preclinical studies have specifically assessed the potential of transplants as an adjunct to the clinical gold standard treatment.” (2)
Interestingly, In 1992, there was a Mark D. Lindner at Bristol-Myers Squibb Pharmaceutical Research Institute (3), and he was still there in 2003 (4).
The most interesting part is, however, that in 1997, a Mark D. Lindner published a paper entitled “Reliability, Distribution, and Validity of Age-Related Cognitive Deficits in the Morris Water Maze,” in which he says both:
“The data presented in this study were collected by the author as a Post-doctoral Research Fellow at Bristol-Myers Squibb Pharmaceutical Research Institute.” (5)
And
“Address correspondence and reprint requests to Mark D. Lindner, CytoTherapeutics, 701 George Washington Highway, Lincoln, RI 02865. E-mail: MLINDNER@CYTO.COM.” (5)
There is no mention of an affiliation with Bristol-Myers Squibb in either of the two papers that use the term "the gold standard."
Bristol-Myers Squibb makes Sinemet (the original levodopa/carbidopa). Sinemet lost patent protection somewhere in the early to mid 90s.
***********************************
(1) Lindner, Mark D., et al. Validation of a Rodent Model of Parkinson's Disease: Evidence of a Therapeutic Window for Oral Sinemet. Brain Research Bulletin, Vol. 39, No. 6, pp. 367-372, 1996 (CytoTherapeutics, Inc., Provicence, RI 02906, USA. MLINDNER@CTYO.COM)
(2) Lindner, Mark D., et al., Somatic delivery of catecholamines in the striatum attenuate parkinsonian symptoms and widen the therapeutic window of oral sinemet in rats. Experimental Neurology, May;145(1):130-40. 1997 (Cyto Therapeutics Inc., Providence, Rhode Island 02906, USA. mlindner@cyto.com)
(3) Lindner MD, Gribkoff VK. Effects of oral BMY 21502 on Morris water task performance in 16-18 month old F-344 rats. Psychopharmacology (Berl). 107(4):485-8, 1992. (Department of Neuropharmacology, Bristol-Myers Squibb Pharmaceutical Research Institute, Wallingford, CT 06492)
(4) Lindner MD, Gribkoff VK, Donlan NA, Jones TA. Long-lasting functional disabilities in middle-aged rats with small cerebral infarcts. J Neurosci. Nov 26;23(34):10913-22, 2003. (Neuroscience Drug Discovery, Bristol-Myers Squibb Pharmaceutical Research Institute, Wallingford, Connecticut 06492, USA. Mark.Lindner@BMS.com)
(5) Lindner, Mark D. Reliability, Distribution, and Validity of Age-Related Cognitive Deficits in the Morris Water Maze. Neurobiology of Learning and Memory, 68, 203–220 1997. (Department of CNS Biology, Bristol-Myers Squibb Pharmaceutical Research Institute)
Monday, October 30, 2006
Monday, October 23, 2006
Rush Didn't Do His Homework
Rush Limbaugh recently opined at how unfortunate it was that Michael J Fox exploits folks by going off his meds for public appearances - he knows that Mr. Fox does this because, you see, "people" have told him. This is my response to Mr. Limbaugh - too long and no, i don't really expect him to take up the challenge, but a girl's gotta try.
******************************
I would recommend doing a little homework before you slap the "exploitative" label on someone because of what "people" have told you.
What you saw Mr. Fox experiencing on the McCaskill ad is a side effect of the most prescribed medication for Parkinson's, levodopa/carbidopa.
Looks pretty bad, right?
If you REALLY wanted to make a difference in people's lives, you might try drawing attention to the fact that this drug has dominated the field of Parkinson's therapies for 36 years - this is a drug that causes a side effect that, when it occurs in the absence of a causal drug, is evidence of neurological disease (Huntington's chorea).
Every single one of the big companies that makes a levodopa product, which causes a plethora of frequently disabling side effects, either makes, or has a generic arm that makes, or is affiliated in some way with a company or companies that make the myriad other drugs that are billed as fixing the problems levodopa causes - pretty neat set up, eh? Sell something that screws people up, then sell them something, and something else, and yet something else, telling them each time that adding THIS drug to the 47 other pills you take every day will surely fix the problem.
Levodopa is generic, you see, so lots of companies rely on that income stream, and the bigger ones have expanded their markets using the means described above – and while current income streams are important, potential for future growth is as, or more, important –and as long as no one breaks the unwritten rule and invents a drug that actually works for Parkinson’s, industry can happily just go on churning out adjunct after adjunct after adjunct – levodopa offers industry infinite potential for growth.
A drug that actually worked destroy all of that. Levodopa has been touted as “the best we have” for 36 years – the best for whom is the question, and the answer is obvious.
If you are REALLY interested in protecting people from exploitation, do something about THAT!
You are no doubt being flooded with emails telling you how badly you screwed up, so for pete’s sake, do the right thing an apologize to Mr. Fox.
******************************
I would recommend doing a little homework before you slap the "exploitative" label on someone because of what "people" have told you.
What you saw Mr. Fox experiencing on the McCaskill ad is a side effect of the most prescribed medication for Parkinson's, levodopa/carbidopa.
Looks pretty bad, right?
If you REALLY wanted to make a difference in people's lives, you might try drawing attention to the fact that this drug has dominated the field of Parkinson's therapies for 36 years - this is a drug that causes a side effect that, when it occurs in the absence of a causal drug, is evidence of neurological disease (Huntington's chorea).
Every single one of the big companies that makes a levodopa product, which causes a plethora of frequently disabling side effects, either makes, or has a generic arm that makes, or is affiliated in some way with a company or companies that make the myriad other drugs that are billed as fixing the problems levodopa causes - pretty neat set up, eh? Sell something that screws people up, then sell them something, and something else, and yet something else, telling them each time that adding THIS drug to the 47 other pills you take every day will surely fix the problem.
Levodopa is generic, you see, so lots of companies rely on that income stream, and the bigger ones have expanded their markets using the means described above – and while current income streams are important, potential for future growth is as, or more, important –and as long as no one breaks the unwritten rule and invents a drug that actually works for Parkinson’s, industry can happily just go on churning out adjunct after adjunct after adjunct – levodopa offers industry infinite potential for growth.
A drug that actually worked destroy all of that. Levodopa has been touted as “the best we have” for 36 years – the best for whom is the question, and the answer is obvious.
If you are REALLY interested in protecting people from exploitation, do something about THAT!
You are no doubt being flooded with emails telling you how badly you screwed up, so for pete’s sake, do the right thing an apologize to Mr. Fox.
Saturday, September 02, 2006
Gambling and Dopamine Agonists: Where's the Beef?
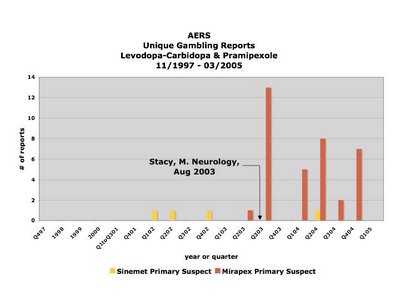
The above is a graph of the distribution over time of the registering of reports linking gambling to either levodopa-carbidopa/Sinemet (yellow) or pramipexole/Mirapex (red) to the FDA's Adverse [Drug] Event Reporting System (AERS) database between November of 1997 and March 2005. The publicizing of the first study purporting to have found an association between pramipexole/Mirapex is denoted in August, 2003, and its effect is clear.
And here is the context for that bar chart.
Ana Szarfman, MD, PhD, P. Murali Doraiswamy, MD, Joseph M. Tonning, MD, MPH, and Jonathan G. Levine, PhD published a research letter to the editor entitled “Association Between Pathologic Gambling and Parkinsonian Therapy as Detected in the Food and Drug Administration Adverse Event Database” in the Archives of Neurology in February of 2006.
In their letter, Szarfman et al say they found, among other things, 39 reports linking Mirapex to gambling in the FDA's AERS database. They then went on to apply the Multi-item Gamma Poisson Shrinker (MGPS) statistical algorithm to the reporting rates of certain drugs in conjunction with gambling. I am not sure how that works, but Mirapex came out the other end of that algorithm looking guiltier than ever.
However, what Szarfman et al failed to reveal was that all but one of the reports in question regarding Mirapex were registered after the first study purporting an association between it and gambling was publicized heavily by the AAN in August of 2003 (Stacy, Driver-Dunckley. Neurology, August 2003).
The fact that Szarfman et al chose not only not to reveal but also not to take what is known as "the publicity effect" into account, particularly one of such magnitude (there was a virtually 100-fold increase in the reporting rate starting immediately after the publicizing of the study in 2003, and this was after extensive clinical trials and six years on the market with only one gambling report) is at odds with a recommendation she and her co-authors make in their 2005 paper entitled “Perspectives on the Use of Data Mining in Pharmacovigilance,” in which they say:
"Publicity resulting from advertising, litigation or regulatory actions (e.g. "Dear healthcare provider" letters and product withdrawals) may result in increased reporting and can generate higher-than-expected relative RRs.[reporting ratios][56,58] Relative RRs should be examined over time in hopes of detecting these influences, although there are no definitive criteria for using data-mining techniques to reliably identify such effects."In this case, the number of reports is so small number and they were registered over such a short period of time that there was no need to use data-mining techniques to detect this effect. In fact, it would be impossible not to see such a sudden skyrocketing in reporting as this, if one were in possession of the data - as I am, having requested it directly from the FDA when Dr. Szarfman declined to provide me with the dates the reports were registered.
Consequently, is difficult to interpret it as anything but disingenuous when Szarfman et al state in their conclusion that the reports stimulated by the reporting of the case series from the 2003 study to the AERS may have increased the MGPS signal scores. It would be difficult to argue against the statement that the stimulated reporting was the MGPS score, and that in fact without it, there would be no MGPS score for Mirapex at all.
If anyone wants to go through the raw data, please contact me – I would be very happy to send it to you – or my refined data – yours for the asking. The dates of the reports don’t lie.
Tuesday, August 29, 2006
Levodopa – if this is the "gold standard," we’re in trouble - Part II
I’ll start with the executive summary, in case you don’t have time to read the whole thing. (Terms are defined in the post following the summary)
Executive Summary
In short, that the last 40 years of research developing different iterations of levodopa were not geared toward improving on its strengths is evident in the incessant singing of its praises (and, since 1996 or so, its donning of the “gold standard” mantle). Rather, the last 40 years of levodopa-centricity have been focused on fixing all the things that are wrong with it.
And, in a nutshell, with respect to levodopa’s deficits, here is my assessment of what has been accomplished since 1970:
The transition from levodopa to Sinemet resulted in a 52% reduction in the incidence of nausea/vomiting, a 60% increase in the incidence of dyskinesias, and had no impact on wearing off or on/off fluctuations.
The transition from Sinemet to Sinemet CR resulted in no improvement in on/off fluctuations, a reduction in the frequency of dosing per day, and a 35% increase in the incidence of dyskinesias.
The transition from Sinemet CR to Stalevo resulted in – if you average the numbers from each of the clinical trials listed in the package insert – about 51 additional minutes of on time per day. It also resulted in a 75% higher incidence of dyskinesias, and that was only after six months.
So for the super duper summary, after 40 years of effort and untold amounts of money wasted trying to put bandaids on this shameful excuse for a “therapy,” the only real improvements that have been achieved are:
The four-fold increase in the incidence of dyskinesias pretty much wipes all of them out.
So – you tell me – has the Parkinson’s disease research community’s obsession with fixing levodopa paid off? [that is the end of the executive summary]
In Part II, each consecutive iteration of levodopa will be examined (4) to see exactly which deficits were targeted for improvement and how well those goals were achieved – an evaluation which requires examination of the effect of the change not only on the targeted deficits, but also on deficits not targeted for improvement.
Because levodopa’s prowess at ameliorating symptoms has never been questioned, I will not be including any examination of the effect of a change on motor scores – any improvements on that front have been gravy; they were not the stated goal of any of the changes, to my knowledge. Therefore, I will focus solely on the impact of 40 years of effort and countless dollars invested in the stated goal – fixing levodopa’s problems.
The milestones I will touch upon consist of either the marrying of levodopa with another drug in the same pill (Sinemet, 1975; and Stalevo, 2003) or actual reformulations of levodopa (Sinemet CR, 1991-2)
Sinemet
Levodopa was approved in 1970 or so. The next iteration in the US was approved in 1975, when Sinemet came on the market. One of the major problems with levodopa on its own was that it frequently caused what was termed “dose-limiting” nausea and vomiting. This was a consequence of the fact that most of the levodopa that was consumed was metabolized before it crossed the blood brain barrier, which meant one had to take what were referred to in one study as “massive oral doses” of the drug to ensure that the tiny percentage that made it to the brain was large enough to have an impact on symptoms. The addition of carbidopa, a decarboxylase inhibitor, to levodopa in 1975 inhibited the metabolizing of levodopa in the digestive tract, which allowed a larger percentage of a smaller dose to reach the brain intact and ready to be metabolized there. This made a big dent in the nausea problem. (1)(5)(6)
However, even way back then, levodopa’s other glaring deficits – dyskinesia (involuntary, uncontrolled muscle movements), wearing off (when the efficacy of the drug wears off before the next dose is due) and on/off fluctuations (when the efficacy of the drug suddenly and unpredictably disappears mid-dose (as opposed to at the end of the dose, which is termed “wearing off”)) - were evident and occurred at an alarmingly high rate – and quickly, too. For example, after one year on levodopa alone, observed rates of dyskinesia ranged from 41% to 60%. (7)(8)
And in their 1975 study, Lieberman et al noted that after two years, dyskinesias were observed in 48% of patients on levodopa, and were present in 77 % of patients on carbidopa/levodopa. That is a 60% increase, just in case you forgot your calculator today. (1)
The increase in dyskinesias might seem at odds with the fact that the decarboxylase inhibitor allowed a 75% reduction in the actual levodopa dose, but it makes perfect sense when one takes into account the fact that in spite of the smaller dose, more levodopa was actually getting to the brain. In fact, the ultimate effect of every improvement to date is that more levodopa gets to the brain.
Sinemet CR
The next improvement came in the form of Sinemet CR. CR stands for controlled release, which means that it is metabolized more slowly, thereby providing a more stable level of the drug in the bloodstream. This reduction of the fluctuation in the level of the drug in the bloodstream was sought after based on the theory that fluctuations in the levodopa plasma level (the amount of levodopa) in the bloodstream might contribute to the development of motor fluctuations (i.e., wearing off and on/off fluctuations). If you only just started reading about Parkinson’s and the latest theories being entertained by the research community, you might be under the impression that this theory (often described using the terms “continuous rather than pulsatile delivery” of the drug) was a relatively new one.
Hardly. The idea that there is an association between the two has been around since at least 1971 (9), and two different controlled release formulations were actually tested in 1978, with differing outcomes (10)(11).
For some reason, it was an additional 13 years before such a formulation was approved for use in the US, and given all that time, and the countless clinical trials undertaken to test out different controlled release formulations, and given that the whole purpose of the endeavor was to decrease the incidence of motor fluctuations, one would think that approval of Sinemet CR would indicate success on that front.
Think again.
The following is an excerpt from Sinemet CR product insert (the sheet of paper, packed with information about the drug, that is supposed to accompany each prescription that is filled) dated 1999:
And as far as dyskinesias go:
Therefore, Sinemet CR, the whole point of which was to ameliorate motor fluctuations, has no effect on/off time (“on time” is when the drug is working and “off time” is when it is not), almost doubles the incidence of on/off fluctuations, and increases the incidence of dyskinesias by over 1/3.
So, while Sinemet CR is not completely without advantages – it does allow for less frequent dosing, after all – in my opinion they are obliterated by its deficits.
Stalevo
The most recent “improvement,” Stalevo (levodopa, carbidopa and entacapone, a COMT inhibitor (another compound that inhibits the metabolism of levodopa)), is also billed as enhancing the stability of the level of levodopa in the blood stream.
And, indeed, according to the package insert, one out of the three clinical trials listed did demonstrate a statistically significant improvement in on/off time (on increased/off decreased by 1.3-1.4 hours) with the addition of a COMT inhibitor. Of course, that means that the other two trials failed to show a statistically significant improvement in actual hours of on-time – though they do manage to squeeze some statistical significance out of the numbers by looking at the percent increase in on-time rather than the actual hours.
However, the incidence of dyskinesias is higher (surprise, surprise), and, in this instance, 75% higher, and the traditional solution to that problem, the lowering of the levodopa dose, failed to ameliorate that side effect in many patients – see the excerpt from the package insert below:
So, if we chart the “improvements” to levodopa, which, as I said, practically by definition were not targeted at its symptomatic benefit, we end up with something like this:
The transition from levodopa to Sinemet resulted in a 52% reduction in the incidence of nausea/vomiting, a 60% increase in the incidence of dyskinesias, and had no impact on wearing off or on/off fluctuations.
The transition from Sinemet to Sinemet CR resulted in no improvement in on/off fluctuations, a reduction in the frequency of dosing per day, and a 35% increase in the incidence of dyskinesias.
The transition from Sinemet CR to Stalevo resulted in – if you average the numbers from each of the clinical trials listed in the package insert – about 51 additional minutes of on time per day. It also resulted in a 75% higher incidence of dyskinesias, and that was only after six months.
So for the super duper summary, after 40 years of effort and untold amounts of money wasted trying to put bandaids on this shameful excuse for a “therapy,” the only real improvements that have been achieved are:
The four-fold increase in the incidence of dyskinesias pretty much wipes all of them out.
In the next installment, I will consider how much money has been thrown at the eternally failing effort to fix levodopa, and how much money is currently being wasted on that same effort. I will also consider the question of whether there is any real incentive, given the size of the PD market, for any pharmaceutical company to risk its capital on a symptomatic therapy that is “better” than levodopa – an undertaking that has been deemed virtually impossible (whoever has been responsible for marketing levodopa has done an excellent job.)
(1) Lieberman A, Goodgold A, Jonas S, Leibowitz M. Comparison of dopa decarboxylase inhibitor (carbidopa) combined with levodopa and levodopa alone in Parkinson's disease. Neurology. 1975 Oct;25(10):911-6. Abstract.
(2) Sinemet CR Product Insert dated 1999.
(3) Stalevo Product Insert dated 2003.
(4) For statistics regarding motor complications with respect to Sinemet CR and Stalevo, the information I put forth is found on the product labeling – seems it would be difficult to argue with that. However, for the time period up to and including Sinemet’s introduction in 1975, my sources are limited to abstracts on PubMed, or the few studies published prior to 1980 I have been able to access on the internet. Studies that old are simply not available online, not even for a price.
(5) Wajsbort J, Dorner A, Wajsbort E. A comparative clinical investigation of the therapeutic effect of levodopa alone and in combination with a decarboxylase inhibitor (carbidopa) in cases of Parkinson's disease. Curr Med Res Opin. 1978;5(9):695-708. Abstract.
(6) Hanzal F. Treatment of Parkinson's syndrome with L-dopa and L-carbidopa. MMW Munch Med Wochenschr. 1976 May 14;118(20):653-6. Abstract.
(7) Markham CH, Treciokas LJ, Diamond SG. Parkinson's disease and levodopa. A five-year follow-up and review. West J Med. 1974 Sep;121(3):188-206.
(8) Shaw KM, Lees AJ, Stern GM. The impact of treatment with levodopa on Parkinson's disease. Q J Med. 1980;49(195):283-93.
(9) Allen JG, Calne DB, Davies CA, Reid JL. Relationship of plasma concentrations of levodopa to clinical response in Parkinsonsim. Br J Pharmacol. 1971 Oct;43(2):464P-465P.
(10) Saarinen A, Myllyla VV, Tokola O, Hokkanen E. Effect of a slow release preparation of levodopa on Parkinson's disease in combination with a peripheral decarboxylase inhibitor. Acta Neurol Scand. 1978 Dec;58(6):340-9. Abstract.
(11) Friedman A. [Preliminary assessment of the effectiveness of L-dopa with prolonged action (Medidopa retard) as compared to conventional L-dopa preparation] [Article in Polish]. Neurol Neurochir Pol. 1978 Jan-Feb;12(1):39-43. Abstract.
Executive Summary
In short, that the last 40 years of research developing different iterations of levodopa were not geared toward improving on its strengths is evident in the incessant singing of its praises (and, since 1996 or so, its donning of the “gold standard” mantle). Rather, the last 40 years of levodopa-centricity have been focused on fixing all the things that are wrong with it.
And, in a nutshell, with respect to levodopa’s deficits, here is my assessment of what has been accomplished since 1970:
The transition from levodopa to Sinemet resulted in a 52% reduction in the incidence of nausea/vomiting, a 60% increase in the incidence of dyskinesias, and had no impact on wearing off or on/off fluctuations.
The transition from Sinemet to Sinemet CR resulted in no improvement in on/off fluctuations, a reduction in the frequency of dosing per day, and a 35% increase in the incidence of dyskinesias.
The transition from Sinemet CR to Stalevo resulted in – if you average the numbers from each of the clinical trials listed in the package insert – about 51 additional minutes of on time per day. It also resulted in a 75% higher incidence of dyskinesias, and that was only after six months.
So for the super duper summary, after 40 years of effort and untold amounts of money wasted trying to put bandaids on this shameful excuse for a “therapy,” the only real improvements that have been achieved are:
- a halving of the incidence of nausea/vomiting (1)
- a reduction in the number of times one has to take medication every day (2)
- and less than an hour of additional on time per day (3)
The four-fold increase in the incidence of dyskinesias pretty much wipes all of them out.
So – you tell me – has the Parkinson’s disease research community’s obsession with fixing levodopa paid off? [that is the end of the executive summary]
In Part II, each consecutive iteration of levodopa will be examined (4) to see exactly which deficits were targeted for improvement and how well those goals were achieved – an evaluation which requires examination of the effect of the change not only on the targeted deficits, but also on deficits not targeted for improvement.
Because levodopa’s prowess at ameliorating symptoms has never been questioned, I will not be including any examination of the effect of a change on motor scores – any improvements on that front have been gravy; they were not the stated goal of any of the changes, to my knowledge. Therefore, I will focus solely on the impact of 40 years of effort and countless dollars invested in the stated goal – fixing levodopa’s problems.
The milestones I will touch upon consist of either the marrying of levodopa with another drug in the same pill (Sinemet, 1975; and Stalevo, 2003) or actual reformulations of levodopa (Sinemet CR, 1991-2)
Sinemet
Levodopa was approved in 1970 or so. The next iteration in the US was approved in 1975, when Sinemet came on the market. One of the major problems with levodopa on its own was that it frequently caused what was termed “dose-limiting” nausea and vomiting. This was a consequence of the fact that most of the levodopa that was consumed was metabolized before it crossed the blood brain barrier, which meant one had to take what were referred to in one study as “massive oral doses” of the drug to ensure that the tiny percentage that made it to the brain was large enough to have an impact on symptoms. The addition of carbidopa, a decarboxylase inhibitor, to levodopa in 1975 inhibited the metabolizing of levodopa in the digestive tract, which allowed a larger percentage of a smaller dose to reach the brain intact and ready to be metabolized there. This made a big dent in the nausea problem. (1)(5)(6)
However, even way back then, levodopa’s other glaring deficits – dyskinesia (involuntary, uncontrolled muscle movements), wearing off (when the efficacy of the drug wears off before the next dose is due) and on/off fluctuations (when the efficacy of the drug suddenly and unpredictably disappears mid-dose (as opposed to at the end of the dose, which is termed “wearing off”)) - were evident and occurred at an alarmingly high rate – and quickly, too. For example, after one year on levodopa alone, observed rates of dyskinesia ranged from 41% to 60%. (7)(8)
And in their 1975 study, Lieberman et al noted that after two years, dyskinesias were observed in 48% of patients on levodopa, and were present in 77 % of patients on carbidopa/levodopa. That is a 60% increase, just in case you forgot your calculator today. (1)
The increase in dyskinesias might seem at odds with the fact that the decarboxylase inhibitor allowed a 75% reduction in the actual levodopa dose, but it makes perfect sense when one takes into account the fact that in spite of the smaller dose, more levodopa was actually getting to the brain. In fact, the ultimate effect of every improvement to date is that more levodopa gets to the brain.
Sinemet CR
The next improvement came in the form of Sinemet CR. CR stands for controlled release, which means that it is metabolized more slowly, thereby providing a more stable level of the drug in the bloodstream. This reduction of the fluctuation in the level of the drug in the bloodstream was sought after based on the theory that fluctuations in the levodopa plasma level (the amount of levodopa) in the bloodstream might contribute to the development of motor fluctuations (i.e., wearing off and on/off fluctuations). If you only just started reading about Parkinson’s and the latest theories being entertained by the research community, you might be under the impression that this theory (often described using the terms “continuous rather than pulsatile delivery” of the drug) was a relatively new one.
Hardly. The idea that there is an association between the two has been around since at least 1971 (9), and two different controlled release formulations were actually tested in 1978, with differing outcomes (10)(11).
For some reason, it was an additional 13 years before such a formulation was approved for use in the US, and given all that time, and the countless clinical trials undertaken to test out different controlled release formulations, and given that the whole purpose of the endeavor was to decrease the incidence of motor fluctuations, one would think that approval of Sinemet CR would indicate success on that front.
Think again.
The following is an excerpt from Sinemet CR product insert (the sheet of paper, packed with information about the drug, that is supposed to accompany each prescription that is filled) dated 1999:
“In clinical trials, patients with moderate to severe motor fluctuations who received SINEMET CR did not experience quantitatively significant reductions in o‘ ff ’time when compared to SINEMET (Carbidopa-Levodopa).” [sic]In fact, according to the table comparing the incidences of adverse experiences with Sinemet CR to those of Sinemet, the incidence of on/off fluctuations (as opposed to amount of time on or off) is almost 50% higher with Sinemet CR than with Sinemet.
And as far as dyskinesias go:
“Carbidopa does not decrease adverse reactions due to central effects of levodopa. By permitting more levodopa to reach the brain, particularly when nausea and vomiting is not a dose-limiting factor, certain adverse CNS effects, e.g., dyskinesias, will occur at lower dosages and sooner during therapy with SINEMET CR (Carbidopa-Levodopa) Sustained-Release than with levodopa alone.” [italicization per original]And:
“Patients receiving SINEMET CR may develop increased dyskinesias compared to SINEMET (Carbidopa-Levodopa).”Indeed, the table comparing the incidences of adverse experiences shows a 35% greater incidence of dyskinesias with Sinemet CR than with Sinemet.
Therefore, Sinemet CR, the whole point of which was to ameliorate motor fluctuations, has no effect on/off time (“on time” is when the drug is working and “off time” is when it is not), almost doubles the incidence of on/off fluctuations, and increases the incidence of dyskinesias by over 1/3.
So, while Sinemet CR is not completely without advantages – it does allow for less frequent dosing, after all – in my opinion they are obliterated by its deficits.
Stalevo
The most recent “improvement,” Stalevo (levodopa, carbidopa and entacapone, a COMT inhibitor (another compound that inhibits the metabolism of levodopa)), is also billed as enhancing the stability of the level of levodopa in the blood stream.
And, indeed, according to the package insert, one out of the three clinical trials listed did demonstrate a statistically significant improvement in on/off time (on increased/off decreased by 1.3-1.4 hours) with the addition of a COMT inhibitor. Of course, that means that the other two trials failed to show a statistically significant improvement in actual hours of on-time – though they do manage to squeeze some statistical significance out of the numbers by looking at the percent increase in on-time rather than the actual hours.
However, the incidence of dyskinesias is higher (surprise, surprise), and, in this instance, 75% higher, and the traditional solution to that problem, the lowering of the levodopa dose, failed to ameliorate that side effect in many patients – see the excerpt from the package insert below:
“Entacapone may potentiate the dopaminergic side effects of levodopa and may therefore cause and/or exacerbate preexisting dyskinesia. Although decreasing the dose of levodopa may ameliorate this side effect, many patients in controlled trials continued to experience frequent dyskinesias despite a reduction in their dose of levodopa.”And that doesn’t even address the problem of how one reduces the levodopa dose alone when levodopa, carbidopa and entacapone are all rolled into one pill.
So, if we chart the “improvements” to levodopa, which, as I said, practically by definition were not targeted at its symptomatic benefit, we end up with something like this:
The transition from levodopa to Sinemet resulted in a 52% reduction in the incidence of nausea/vomiting, a 60% increase in the incidence of dyskinesias, and had no impact on wearing off or on/off fluctuations.
The transition from Sinemet to Sinemet CR resulted in no improvement in on/off fluctuations, a reduction in the frequency of dosing per day, and a 35% increase in the incidence of dyskinesias.
The transition from Sinemet CR to Stalevo resulted in – if you average the numbers from each of the clinical trials listed in the package insert – about 51 additional minutes of on time per day. It also resulted in a 75% higher incidence of dyskinesias, and that was only after six months.
So for the super duper summary, after 40 years of effort and untold amounts of money wasted trying to put bandaids on this shameful excuse for a “therapy,” the only real improvements that have been achieved are:
- a halving of the incidence of nausea/vomiting
- a reduction in the number of times one has to take medication every day
- and less than an hour of additional on time per day
The four-fold increase in the incidence of dyskinesias pretty much wipes all of them out.
In the next installment, I will consider how much money has been thrown at the eternally failing effort to fix levodopa, and how much money is currently being wasted on that same effort. I will also consider the question of whether there is any real incentive, given the size of the PD market, for any pharmaceutical company to risk its capital on a symptomatic therapy that is “better” than levodopa – an undertaking that has been deemed virtually impossible (whoever has been responsible for marketing levodopa has done an excellent job.)
(1) Lieberman A, Goodgold A, Jonas S, Leibowitz M. Comparison of dopa decarboxylase inhibitor (carbidopa) combined with levodopa and levodopa alone in Parkinson's disease. Neurology. 1975 Oct;25(10):911-6. Abstract.
(2) Sinemet CR Product Insert dated 1999.
(3) Stalevo Product Insert dated 2003.
(4) For statistics regarding motor complications with respect to Sinemet CR and Stalevo, the information I put forth is found on the product labeling – seems it would be difficult to argue with that. However, for the time period up to and including Sinemet’s introduction in 1975, my sources are limited to abstracts on PubMed, or the few studies published prior to 1980 I have been able to access on the internet. Studies that old are simply not available online, not even for a price.
(5) Wajsbort J, Dorner A, Wajsbort E. A comparative clinical investigation of the therapeutic effect of levodopa alone and in combination with a decarboxylase inhibitor (carbidopa) in cases of Parkinson's disease. Curr Med Res Opin. 1978;5(9):695-708. Abstract.
(6) Hanzal F. Treatment of Parkinson's syndrome with L-dopa and L-carbidopa. MMW Munch Med Wochenschr. 1976 May 14;118(20):653-6. Abstract.
(7) Markham CH, Treciokas LJ, Diamond SG. Parkinson's disease and levodopa. A five-year follow-up and review. West J Med. 1974 Sep;121(3):188-206.
(8) Shaw KM, Lees AJ, Stern GM. The impact of treatment with levodopa on Parkinson's disease. Q J Med. 1980;49(195):283-93.
(9) Allen JG, Calne DB, Davies CA, Reid JL. Relationship of plasma concentrations of levodopa to clinical response in Parkinsonsim. Br J Pharmacol. 1971 Oct;43(2):464P-465P.
(10) Saarinen A, Myllyla VV, Tokola O, Hokkanen E. Effect of a slow release preparation of levodopa on Parkinson's disease in combination with a peripheral decarboxylase inhibitor. Acta Neurol Scand. 1978 Dec;58(6):340-9. Abstract.
(11) Friedman A. [Preliminary assessment of the effectiveness of L-dopa with prolonged action (Medidopa retard) as compared to conventional L-dopa preparation] [Article in Polish]. Neurol Neurochir Pol. 1978 Jan-Feb;12(1):39-43. Abstract.
Friday, July 28, 2006
Levodopa – if this is the “gold standard,” we are in trouble
Parkinson’s disease (PD) is a degenerative neurological condition in which neurons in the brain that produce dopamine, a neurotransmitter that facilitates control and modulation of muscle movement, slowly die. Tremor is the symptom most commonly associated with PD in people’s minds, but it is the other two cardinal symptoms, rigidity and bradykinesia (slowness of movement (1)), that bring people with Parkinson’s (PWP) to a halt.
Described in that way, it sounds rather benign, doesn’t it – it is not. It doesn’t just rob one of niceties like being able to write with pen and paper, or dance, or play the piano, or type, or cut one’s food at dinner. Increasing disability forces people to quit working and try to survive on disability payments. Showering, shaving, getting dressed and eating become more and more challenging. People fall and break arms, legs, teeth. Sometimes they fall and can’t get up – for days, potentially, if they live alone – rigidity can be that severe. People end up in wheelchairs. Incontinence can occur, though I do not know with what frequency because I don't want to know. Swallowing can become a problem, which can lead to aspiration, pneumonia, and worse.
Given enough time, PD will result in disabling immobility and, ultimately, death, if nothing else gets you first.
In 1970 or so, the drug levodopa was introduced to treat PD. At first it appeared to be a miracle drug – people who had been immobilized by the disease were able to move again. However, even during clinical trials its most visible side effect, dyskinesias (uncontrollable, involuntary muscle movements) had become evident, and certainly by 1980, the research community was well aware the levodopa not only caused dyskinesias, but it also lost efficacy very quickly.
A study published in 1980 by Shaw, et. al. followed 178 patients who were evaluated at various points between the start of levodopa therapy, which occurred sometime between November 1969 and December 1972, and the final evaluation in 1977. (2)
Chart One (click chart for larger view)

Of the original 178 patients, only 81 made it through to six years on levodopa. 70 dropped out, most of them before the two-year mark, due to severity of side effects. An additional 27 were lost to follow-up, either because of death or unknown whereabouts.
Shaw et. al. categorize the benefit derived from levodopa as marked (50-100% improvement in rating score), moderate (25-50% improvement), slight (0-25% improvement) or none, and as you can see below, only 20%, or 36, of the original 178 were still taking levodopa and deriving moderate benefit at the six-year mark – and that was the best that anyone was doing – there was no one in the “markedly improved” category.
Chart Two (click chart for larger view)

As a 42 year old with PD, I don’t find that particularly encouraging, nor would I if I were 60, which is often cited as the average age of onset (though I have seen 50 cited, as well.) And that is only half the story. Levodopa has some pretty devastating side effects. Consider rates of occurrence of three of them in the chart below, as found in the Shaw study mentioned above.
Chart Three (click chart for larger view)

We all know what dementia is. “Wearing off” is a euphemism for loss of efficacy – it refers to the phenomenon in which over time, the length of time each dose is effective diminishes. After a few years, it is quite common for people to be medicating every two hours, and even then, the medication may “wear off” before the next dose is due, leaving them coping with some very dangerous and incapacitating symptoms.
Dyskinesias, as noted above, are involuntary, uncontrollable muscle movements. It is difficult to convey to one who has never seen them exactly what they look like and how disabling they can be, so I found a description of severe dyskinesias in a study.
I do not have dyskinesias myself, but have come into contact with quite a few people who do. I saw one Parkinson’s patient whose body was in such motion that she was unable to simply pick up a glass and drink from it. Instead, she requested a straw, which allowed her to sip from it while leaving it on the table. I have spoken to people on the phone whose movements were so vigorous that I could hear the difficulty they were having keeping the handset positioned correctly. I spoke to one woman on the phone who said she was lying on the floor because her bodily movements were so violent that she could neither stand nor sit; I could barely hear her over the noise of the handset being knocked around. I witnessed a man whose involuntary writhing was so intense that he could barely keep from sliding off his chair onto the floor. At a fundraising dinner one night, I saw a woman whose head was flinging back and forth so wildly it was a miracle she didn’t smash it on the table.
And here’s the kicker; according to Shaw, et. al.:
Excellent! Not only was there only a mere 20% chance of still deriving, at best, moderate benefit from levodopa at the six-year mark, and not only was there about an 100% chance that one would be dealing with dyskinesias, but the moderate benefit would come when the dyskinesias were the worst. Oh, and let’s not forget the dangers of dementia and wearing off.
Given the above, I have to say that I don’t quite get it when Shaw, et. al. say:
Most didn’t even make it to the six year mark! 35% dropped out by the second year! Only 45% actually finished the study and only 20% finished the study at a moderate level of benefit, and that was being compromised by concomitant dyskinesias! And that was the best case scenario – the rest of the people who finished the study were experiencing slight to zero benefit but were almost certainly experiencing the joys of dyskinesias and wearing off, and if they got really lucky, dementia, too!
Unfortunately, such compartmentalization of the benefits and deficits of levodopa is standard. In cases in which a side effect may be troubling (i.e., dry mouth) but not interfere with the actual benefit being derived from the drug (e.g., the ability to move, and thereby accomplish tasks requiring movement, e.g., picking up and drinking from a glass of water), it might not be misleading to compartmentalize the benefit and present it as separate and distinct from the deficit. However, when the benefit itself is compromised by the side effect, it does become misleading to present them as separate and unrelated, as is almost always done
Levodopa may be better than nothing – I do not have enough information to comment on that designation – but to call it “the gold standard,” as has been done for many years, conveys the profoundly inaccurate impression that this drug is better than good enough by cavalierly disregarding side effects of such severity and impact that in any other field of medicine would be considered evidence of toxicity.
I have provided references below and I encourage anyone and everyone to take a look at them. My rational for calculating incidences a little differently than the authors of the Shaw study did is elucidated in footnote (4).
My next entry will focus on a more recent study to explore the degree to which the billions of dollars that have been invested in trying to fix levodopa over the last 40 or so years have succeeded.
* From Chart Three: The authors report incidences of three different kinds of dyskinesias in the group that made it to the final evaluation – peak dose, 80%; end of dose dystonia, 20%; and biphasic, 3%. Clearly, there is overlap somewhere, but since it is not elucidated, I have chosen to aggregate the percentages rather than try to guess how they might overlap.
Described in that way, it sounds rather benign, doesn’t it – it is not. It doesn’t just rob one of niceties like being able to write with pen and paper, or dance, or play the piano, or type, or cut one’s food at dinner. Increasing disability forces people to quit working and try to survive on disability payments. Showering, shaving, getting dressed and eating become more and more challenging. People fall and break arms, legs, teeth. Sometimes they fall and can’t get up – for days, potentially, if they live alone – rigidity can be that severe. People end up in wheelchairs. Incontinence can occur, though I do not know with what frequency because I don't want to know. Swallowing can become a problem, which can lead to aspiration, pneumonia, and worse.
Given enough time, PD will result in disabling immobility and, ultimately, death, if nothing else gets you first.
In 1970 or so, the drug levodopa was introduced to treat PD. At first it appeared to be a miracle drug – people who had been immobilized by the disease were able to move again. However, even during clinical trials its most visible side effect, dyskinesias (uncontrollable, involuntary muscle movements) had become evident, and certainly by 1980, the research community was well aware the levodopa not only caused dyskinesias, but it also lost efficacy very quickly.
A study published in 1980 by Shaw, et. al. followed 178 patients who were evaluated at various points between the start of levodopa therapy, which occurred sometime between November 1969 and December 1972, and the final evaluation in 1977. (2)
Chart One (click chart for larger view)

Of the original 178 patients, only 81 made it through to six years on levodopa. 70 dropped out, most of them before the two-year mark, due to severity of side effects. An additional 27 were lost to follow-up, either because of death or unknown whereabouts.
Shaw et. al. categorize the benefit derived from levodopa as marked (50-100% improvement in rating score), moderate (25-50% improvement), slight (0-25% improvement) or none, and as you can see below, only 20%, or 36, of the original 178 were still taking levodopa and deriving moderate benefit at the six-year mark – and that was the best that anyone was doing – there was no one in the “markedly improved” category.
Chart Two (click chart for larger view)

As a 42 year old with PD, I don’t find that particularly encouraging, nor would I if I were 60, which is often cited as the average age of onset (though I have seen 50 cited, as well.) And that is only half the story. Levodopa has some pretty devastating side effects. Consider rates of occurrence of three of them in the chart below, as found in the Shaw study mentioned above.
Chart Three (click chart for larger view)

We all know what dementia is. “Wearing off” is a euphemism for loss of efficacy – it refers to the phenomenon in which over time, the length of time each dose is effective diminishes. After a few years, it is quite common for people to be medicating every two hours, and even then, the medication may “wear off” before the next dose is due, leaving them coping with some very dangerous and incapacitating symptoms.
Dyskinesias, as noted above, are involuntary, uncontrollable muscle movements. It is difficult to convey to one who has never seen them exactly what they look like and how disabling they can be, so I found a description of severe dyskinesias in a study.
At approximately 7:30 PM [each evening], he began to have violent flinging movements of the arms and legs and facial grimacing interrupted by severe dystonic spasms of the right limbs. He could only attempt to lie down but would flail his entire body from side to side and could not remain in bed due to the violence of the movements. These severe dyskinesias lasted until 11 PM or midnight. (3)
I do not have dyskinesias myself, but have come into contact with quite a few people who do. I saw one Parkinson’s patient whose body was in such motion that she was unable to simply pick up a glass and drink from it. Instead, she requested a straw, which allowed her to sip from it while leaving it on the table. I have spoken to people on the phone whose movements were so vigorous that I could hear the difficulty they were having keeping the handset positioned correctly. I spoke to one woman on the phone who said she was lying on the floor because her bodily movements were so violent that she could neither stand nor sit; I could barely hear her over the noise of the handset being knocked around. I witnessed a man whose involuntary writhing was so intense that he could barely keep from sliding off his chair onto the floor. At a fundraising dinner one night, I saw a woman whose head was flinging back and forth so wildly it was a miracle she didn’t smash it on the table.
And here’s the kicker; according to Shaw, et. al.:
“Movements were most severe when the patient was obtaining maximal relief from rigidity and akinesia…”
Excellent! Not only was there only a mere 20% chance of still deriving, at best, moderate benefit from levodopa at the six-year mark, and not only was there about an 100% chance that one would be dealing with dyskinesias, but the moderate benefit would come when the dyskinesias were the worst. Oh, and let’s not forget the dangers of dementia and wearing off.
Given the above, I have to say that I don’t quite get it when Shaw, et. al. say:
“Despite the unquestionable diminution of responsiveness to levodopa over the years and associated long-term adverse reactions, the quality of life for the average patient treated with levodopa has clearly improved considerably. For two to three years the majority do well, many patients remain gainfully employed and lead active lives for several more years and even even after six years most are still gaining benefit.”
Most didn’t even make it to the six year mark! 35% dropped out by the second year! Only 45% actually finished the study and only 20% finished the study at a moderate level of benefit, and that was being compromised by concomitant dyskinesias! And that was the best case scenario – the rest of the people who finished the study were experiencing slight to zero benefit but were almost certainly experiencing the joys of dyskinesias and wearing off, and if they got really lucky, dementia, too!
Unfortunately, such compartmentalization of the benefits and deficits of levodopa is standard. In cases in which a side effect may be troubling (i.e., dry mouth) but not interfere with the actual benefit being derived from the drug (e.g., the ability to move, and thereby accomplish tasks requiring movement, e.g., picking up and drinking from a glass of water), it might not be misleading to compartmentalize the benefit and present it as separate and distinct from the deficit. However, when the benefit itself is compromised by the side effect, it does become misleading to present them as separate and unrelated, as is almost always done
Levodopa may be better than nothing – I do not have enough information to comment on that designation – but to call it “the gold standard,” as has been done for many years, conveys the profoundly inaccurate impression that this drug is better than good enough by cavalierly disregarding side effects of such severity and impact that in any other field of medicine would be considered evidence of toxicity.
I have provided references below and I encourage anyone and everyone to take a look at them. My rational for calculating incidences a little differently than the authors of the Shaw study did is elucidated in footnote (4).
My next entry will focus on a more recent study to explore the degree to which the billions of dollars that have been invested in trying to fix levodopa over the last 40 or so years have succeeded.
- Bradykinesia: imagine you have been shoveling without gloves and your hands are really cold, and that you are now trying to get your keys out of your pocket and unlock the door – that is what bradykinesia feels like, only without the coldness - at least to me.
- The Impact of Treatment with Levodopa on Parkinson's Disease. K.M. Shaw, A.J. Shaw & G.M. Stern. Quarterly Journal of Medicine, New Series XLIX, No. 195, pp. 283-293, Summer 1980.
- Motor Response to Levodopa and the Evolution of Motor Fluctuations in the First Decade of Treatment of Parkinson’s Disease. CD McColl et. al. Movement Disorders, Vol 17 No 6. 2002. P,, 1227-1234.
- The authors of the study calculate the incidences of the different levels of benefit being derived at six years based only on the population that actually finished the study (n=81). However, since what is important to me as a person with PD is if I start taking levodopa now, what are the odds that it will continue to provide sufficient benefit up to and including the six year mark – in which case the fact that 70 out of 178 people dropped out due to severity of side effects is 100% relevant, and those 70 people are made irrelevant in the authors’ calculations. Therefore, I use the entire study population (n=178) as the denominator when calculating the likelihood of a certain degree of benefit at six years.
However, with respect to side effects, I want to know what my odds are if making it to six years is taken as a given – and so for that calculation I use only the population completing the study (n=81) as the denominator.
* From Chart Three: The authors report incidences of three different kinds of dyskinesias in the group that made it to the final evaluation – peak dose, 80%; end of dose dystonia, 20%; and biphasic, 3%. Clearly, there is overlap somewhere, but since it is not elucidated, I have chosen to aggregate the percentages rather than try to guess how they might overlap.
Sunday, July 23, 2006
Adult Stem Cells NOT Treating Parkinson's and 36 Other Diseases on Brownback's List
Here is a comment that was made by "bmmg39" on my Michael Fumento letter; it is followed by my response:
"Anonymous, no one has claimed that ASCs are "curing" Parkinson's and other diseases. They are successfully TREATING people with those diseases, something ESCs have yet to do for even one human patient. By the way, Anuket, HUMAN ESCs were only isolated in 1998, but non-human ones have been studied and experimented on for decades. You can't complain about a so-called head start."
Oh my goodness, bmmg39, they are NOT successfully or in any other capacity treating Parkinson’s with ASCs. Call The Michael J Fox Foundation, Parkinson’s Disease Foundation, National Parkinson Foundation, American Parkinson’s Disease Foundation – call one, call ‘em all – check their websites – I guarantee you, you will find nothing – there is one person on this planet who has received an autolgous stem cell transplant for Parkinson’s, and that was over five years ago. So, if ASCs are being used to treat (as in FDA-approved treatment, not one person in a clinical trial) people with Parkinson’s, someone is doing a very good job of hiding it from us. If you are in on the secret, please have mercy on us and tell us where to go to get this treatment.
As far as all of the other diseases allegedly being treated or cured with ASCs, I have done the footwork – have you? Have you looked up each and every disease/condition on, for example, Senator Brownback’s list? Well, neither have I, because I got sick of it, but I did look up 62 out of 69 of them, and ASCs are not listed by NIH as a treatment for 36 out of 62 of the diseases on Brownback’s list. No doubt ASCs are being studied in relation to some of the 36, but something that is in clinical trials is by definition not yet a treatment.
Of the 26 diseases/conditions actually being treated with ASCs, I can only see one (testicular cancer) that isn’t a disease of the blood – and those have been being treated for the last 40 years with bone marrow transplants – nothing new there.
And regarding the headstart – if you are going to count ECS animal experiments, I’m afraid you have to count them for ASCs, too. When do you think they started? The first listing I find in PubMed that references the species on which the experiment was performed was in 1955 – guinea pig (PMID: 13344491). The very first bone marrow transplant (BMT) study in PubMed is dated 1950 (PMID: 15442952). That far back, all that is provided is the title of the study, and this one does not reveal the species, but you can be sure that if someone was working on guinea pigs in 1955, no one was working with humans in 1950.
The first animal ESCs, however, were not isolated until 1981 (see this month's Nature). So even if we take 1950 as the beginning of ASC animal studies (they undoubtedly started earlier) there is a minimum of a 30-year gap. I consider that a bit of a head start, don’t you?
It would be helpful if more people did just a little more footwork rather than just blindly believing words because they want them to be true.
"Anonymous, no one has claimed that ASCs are "curing" Parkinson's and other diseases. They are successfully TREATING people with those diseases, something ESCs have yet to do for even one human patient. By the way, Anuket, HUMAN ESCs were only isolated in 1998, but non-human ones have been studied and experimented on for decades. You can't complain about a so-called head start."
Oh my goodness, bmmg39, they are NOT successfully or in any other capacity treating Parkinson’s with ASCs. Call The Michael J Fox Foundation, Parkinson’s Disease Foundation, National Parkinson Foundation, American Parkinson’s Disease Foundation – call one, call ‘em all – check their websites – I guarantee you, you will find nothing – there is one person on this planet who has received an autolgous stem cell transplant for Parkinson’s, and that was over five years ago. So, if ASCs are being used to treat (as in FDA-approved treatment, not one person in a clinical trial) people with Parkinson’s, someone is doing a very good job of hiding it from us. If you are in on the secret, please have mercy on us and tell us where to go to get this treatment.
As far as all of the other diseases allegedly being treated or cured with ASCs, I have done the footwork – have you? Have you looked up each and every disease/condition on, for example, Senator Brownback’s list? Well, neither have I, because I got sick of it, but I did look up 62 out of 69 of them, and ASCs are not listed by NIH as a treatment for 36 out of 62 of the diseases on Brownback’s list. No doubt ASCs are being studied in relation to some of the 36, but something that is in clinical trials is by definition not yet a treatment.
Of the 26 diseases/conditions actually being treated with ASCs, I can only see one (testicular cancer) that isn’t a disease of the blood – and those have been being treated for the last 40 years with bone marrow transplants – nothing new there.
And regarding the headstart – if you are going to count ECS animal experiments, I’m afraid you have to count them for ASCs, too. When do you think they started? The first listing I find in PubMed that references the species on which the experiment was performed was in 1955 – guinea pig (PMID: 13344491). The very first bone marrow transplant (BMT) study in PubMed is dated 1950 (PMID: 15442952). That far back, all that is provided is the title of the study, and this one does not reveal the species, but you can be sure that if someone was working on guinea pigs in 1955, no one was working with humans in 1950.
The first animal ESCs, however, were not isolated until 1981 (see this month's Nature). So even if we take 1950 as the beginning of ASC animal studies (they undoubtedly started earlier) there is a minimum of a 30-year gap. I consider that a bit of a head start, don’t you?
It would be helpful if more people did just a little more footwork rather than just blindly believing words because they want them to be true.
Monday, March 27, 2006
Open Letter to Michael Fumento
Michael Fumento wants his readers to think adult stem cell research (ASCR) is the underdog soooooo badly, it is almost cute. He needs ASCR to be the underdog, of course, to create the illusion that those of us who support embryonic stem cell research (ESCR) are the enemies of ASCR - then he can paint us as trying to hide ASCR successes and steal government funding for ASCR - you know, it is that whole "don't have an enemy? need one? what are you waiting for? create one!"
The reality, of course, is that supporters of ESCR are neither stealing government funding away fron ASCR, nor are we hiding ASCRs successes - many of us would stand to benefit from any ASCR gains - does he think we are nuts? No, of course he doesn't. He just wants the rest of the world to think we are.
However, as I once heard someone say, read it and weep, Mr. Fumento. You know the answers to the questions below as well as I do, so step up to the truth, if you dare. [answers provided by anuket 7/29/06]
-------------------------------------
Hi Michael,
My name is Anuket, and I have some questions about the stem cell issue that I wonder if you would answer publicly so that others can be enlightened, as well. I guess I will just list them.
Thank you for your time and I look forward to reading your response, and I hope you will post the answers to these questions on your site.
(I am calling this an open letter but am not sure it really is one, since I already sent these questions to Mr. Fumento privately, asking him to answer them publicly. He declined to answer them at all.)
The reality, of course, is that supporters of ESCR are neither stealing government funding away fron ASCR, nor are we hiding ASCRs successes - many of us would stand to benefit from any ASCR gains - does he think we are nuts? No, of course he doesn't. He just wants the rest of the world to think we are.
However, as I once heard someone say, read it and weep, Mr. Fumento. You know the answers to the questions below as well as I do, so step up to the truth, if you dare. [answers provided by anuket 7/29/06]
-------------------------------------
Hi Michael,
My name is Anuket, and I have some questions about the stem cell issue that I wonder if you would answer publicly so that others can be enlightened, as well. I guess I will just list them.
- How much funding has adult stem cell research (ASCR) received from the federal government since 2003, and how much is it budgeted to receive by the end of 2007?
2003 to 2005 actual: $593M
2006 to 2007 budgeted: $400M
TOTAL PROJECTED 2003 to 2007: $993M - How much funding has embryonic stem cell research (ESCR) received from the federal government since 2003, and how much is it budgeted to receive by the end of 2007?
2003 to 2005 actual: $84M
2006 to 2007 budgeted: $77M
TOTAL PROJECTED 2003 to 2007: $161M
Numbers taken from the NIH table of Disease Funding Levels - What did Clinton do about ESCR? Did he fund any? Did he do anything?
On August 25, 2000, the Clinton Administration's NIH Guidelines for Research Using Human Pluripotent Stem Cells were printed in the Federal Register [Page 51975], lifting the moratorium on research using human pluripotent stem cells derived from human embryos and fetal tissue put in place by the Director, NIH, in January, 1999. These guidelines authorized federal funding for research utilizing ESCs that were derived from embryos donated by people undergoing IVF. The actual derivation of the ESCs would have to be done using private funding, but once the cell line was created, researchers could obtain NIH funding to work with them.
The first meeting of the NIH committee charge with reviewing grant applications for ESC research was scheduled for April, 2001. Bush took office in January of 2001 and ordered that the Clinton policy be reviewed - the April meeting was cancelled.
In August of 2001, Bush announced the current policy. - How long has human ASCR been going on?
The first studies utilizing human ASCs listed in Pubmed are in the early to mid-60s, depending on the search term used (stem cells, adult stem cells, bone marrow transplant), so human ASCR has been underway for a minimum of 40 years. - How long has human ESCR been going on?
Human embryonic stem cells were first isolated in 1998, so human ESCR has been underway for about seven years. - On the list of diseases being treated with ASCs that you link to:
- How many of these diseases are diseases of the blood, or the immune system, or metabolic disorders?
- How many of these treatments consist of bone marrow transplants?
- Would you define "treat" for me? Do you mean an ASC therapy was tried in a clinical trial and seemed to have a positive outcome, but the therapy is not yet approved by the FDA and available to us, or do you mean a therapy that has been approved by the FDA and is readily available to us?
- How many of the diseases listed are neurological in nature?
- How many types of adult stem cells are currently being used in therapies that have been approved by the FDA and are available to patients today?
- My random sampling of 7 disease turned up 2 that, according to NIH, are not being treated with ASCs - how many of these diseases are actually not being treated with ASCs?
- How long do you estimate it takes, on average, with a new idea for a therapy, to get from the lab, through animal trials, to the first human clinical trial?
- The embryos in question - the ones that we are talking about when we say ESCR - who donates them to research?
People undergoing IVF donate them to research. - Should the people who donate them to research have the right to do that?
- Do you oppose IVF?
Thank you for your time and I look forward to reading your response, and I hope you will post the answers to these questions on your site.
(I am calling this an open letter but am not sure it really is one, since I already sent these questions to Mr. Fumento privately, asking him to answer them publicly. He declined to answer them at all.)
Sunday, February 05, 2006
DoNoHarm: Is It Lies or Is It..... um, Lies...
I sent the following letter to five of the seven esteemed founders of the site DoNoHarm today. The founders are:
Kevin FitzGerald, SJ, PhD
C. Christopher Hook, MD
Ralph Miech, MD, PhD
Robert D. Orr, MD
David A. Prentice, PhD
Frank E. Young, MD, PhD
Joseph Zanga, MD, FAAP
Click here to see the "fact" sheet in question, and click here to see the studies cited to support the whacked out assertion that adult stem cells are being used to treat Parkinson's.
Dear Sirs,
I am writing to make you aware of gross inaccuracies in the Parkinson’s “fact” sheet on the DoNoHarm site and to request that Parkinson's be removed from the site's list of disease being treated with adult stem cells, as it is not being treated with adult stem cells.
The ESC (embryonic stem cell) research section is actually not bad. The five studies cited a) were actually published, b) had a suitable number of subjects, c) actually used ESCs, and d) resulted in improvement, benefit, and alleviation of symptoms to some degree in each and every instance. There are always obstacles that need to be resolved when an area of medical research is in its infancy, as ESC research is, and it is actually quite encouraging that tumors only developed in two of the five studies listed.
However, the ASCs (adult stem cell) section is all wrong, and I mean that quite literally. Of the seven items listed, four do not utilize ASCs at all, one cannot be said to be a study, never mind a treatment, and one cannot be verified at all as it is not clear it has been published. The seventh item actually is a published study, and it actually used ASCs, but being a study cannot be put forth as supporting a claim of “”treatment,” not to mention the fact that the study concludes that more study is necessary.
I will be more specific.
The 2003 study by Gill examined the effect of a neurotrophic factor - GDNF, to be exact - on people with Parkinson’s. The 2005 pathology follow-up to the Gill study by S. Love revealed that the brain treated with GDNF showed signs of neuronal sprouting. GDNF is not ASCs. Likewise, the 2000 Fallon study utilized a “growth protein,” not ASCs.
Further, the 2002 Akerud study did not use ASCs, either. As it says in the study, “To stably deliver PSP in vivo we engineered a c17.2 NSC line,” and the C17.2 NSC (neuronal stem cell) line was derived from “neonatal mouse cerebellar external germinal zone cells.” (jvi.asm.org) Now, technically, the neuronal stem cells are not embryonic, but somehow I don’t think any of you would condone the use of neonatal human cerebellar external germinal zone cells, either - would you? [It is with great embarrassment that I add, after the fact, that this was an animal study, to boot.]
These studies should be removed from the “fact” sheet first of all because they do not even evaluate ASCs for their therapeutic potential, never mind constituting ASC-based "treatments" for Parkinson's.
The inclusion of Dr. Levesque’s work is problematic on several levels. First, to date, Dr. Levesque has merely presented his work at a conference - he has not published his completed study results in a peer-reviewed journal. Secondly, the results he presented to the conference (his only known results) were based on observations of a single subject - as all of you are well aware, one subject is not a meaningful data source. Moreover, because there was only one subject, the study could not have been double blind, placebo (or sham surgery) controlled. Each of these concerns taken on its own is cause for removing this information from the ”fact” sheet.
Taken together, the inclusion this information as a “treatment” in the “fact” sheet is baffling. This information should be removed.
I am unable to confirm the information said to have been presented to the Japan Neurological Society by Okayama University in 2001, as the only corroboration provided is a newspaper article, as opposed to, say, a study published in a peer-reviewed journal. This information should be removed.
Thus, the 2004 Mari Dezawa study is the only item that actually used ASCs on a meaningful number of subjects and was published in a peer-reviewed journal. However, given that it was a study, and especially given that it concluded that “further studies are needed...to ensure the long-term safety and efficacy of manipulated MSCs,” to call it evidence of ASCs “treating” Parkinson’s disease is grossly premature. [Again, it is with great embarrassment that I add, after the fact, that this, too, was an animal study.]
The fact of the matter is that there are no treatments for Parkinson’s utilizing ASCs. I wish there were. But until there are, if credibility is at all important to you, you will make sure that all the DoNoHarm's claims to that effect are removed from the site.
Kevin FitzGerald, SJ, PhD
C. Christopher Hook, MD
Ralph Miech, MD, PhD
Robert D. Orr, MD
David A. Prentice, PhD
Frank E. Young, MD, PhD
Joseph Zanga, MD, FAAP
Click here to see the "fact" sheet in question, and click here to see the studies cited to support the whacked out assertion that adult stem cells are being used to treat Parkinson's.
Dear Sirs,
I am writing to make you aware of gross inaccuracies in the Parkinson’s “fact” sheet on the DoNoHarm site and to request that Parkinson's be removed from the site's list of disease being treated with adult stem cells, as it is not being treated with adult stem cells.
The ESC (embryonic stem cell) research section is actually not bad. The five studies cited a) were actually published, b) had a suitable number of subjects, c) actually used ESCs, and d) resulted in improvement, benefit, and alleviation of symptoms to some degree in each and every instance. There are always obstacles that need to be resolved when an area of medical research is in its infancy, as ESC research is, and it is actually quite encouraging that tumors only developed in two of the five studies listed.
However, the ASCs (adult stem cell) section is all wrong, and I mean that quite literally. Of the seven items listed, four do not utilize ASCs at all, one cannot be said to be a study, never mind a treatment, and one cannot be verified at all as it is not clear it has been published. The seventh item actually is a published study, and it actually used ASCs, but being a study cannot be put forth as supporting a claim of “”treatment,” not to mention the fact that the study concludes that more study is necessary.
I will be more specific.
The 2003 study by Gill examined the effect of a neurotrophic factor - GDNF, to be exact - on people with Parkinson’s. The 2005 pathology follow-up to the Gill study by S. Love revealed that the brain treated with GDNF showed signs of neuronal sprouting. GDNF is not ASCs. Likewise, the 2000 Fallon study utilized a “growth protein,” not ASCs.
Further, the 2002 Akerud study did not use ASCs, either. As it says in the study, “To stably deliver PSP in vivo we engineered a c17.2 NSC line,” and the C17.2 NSC (neuronal stem cell) line was derived from “neonatal mouse cerebellar external germinal zone cells.” (jvi.asm.org) Now, technically, the neuronal stem cells are not embryonic, but somehow I don’t think any of you would condone the use of neonatal human cerebellar external germinal zone cells, either - would you? [It is with great embarrassment that I add, after the fact, that this was an animal study, to boot.]
These studies should be removed from the “fact” sheet first of all because they do not even evaluate ASCs for their therapeutic potential, never mind constituting ASC-based "treatments" for Parkinson's.
The inclusion of Dr. Levesque’s work is problematic on several levels. First, to date, Dr. Levesque has merely presented his work at a conference - he has not published his completed study results in a peer-reviewed journal. Secondly, the results he presented to the conference (his only known results) were based on observations of a single subject - as all of you are well aware, one subject is not a meaningful data source. Moreover, because there was only one subject, the study could not have been double blind, placebo (or sham surgery) controlled. Each of these concerns taken on its own is cause for removing this information from the ”fact” sheet.
Taken together, the inclusion this information as a “treatment” in the “fact” sheet is baffling. This information should be removed.
I am unable to confirm the information said to have been presented to the Japan Neurological Society by Okayama University in 2001, as the only corroboration provided is a newspaper article, as opposed to, say, a study published in a peer-reviewed journal. This information should be removed.
Thus, the 2004 Mari Dezawa study is the only item that actually used ASCs on a meaningful number of subjects and was published in a peer-reviewed journal. However, given that it was a study, and especially given that it concluded that “further studies are needed...to ensure the long-term safety and efficacy of manipulated MSCs,” to call it evidence of ASCs “treating” Parkinson’s disease is grossly premature. [Again, it is with great embarrassment that I add, after the fact, that this, too, was an animal study.]
The fact of the matter is that there are no treatments for Parkinson’s utilizing ASCs. I wish there were. But until there are, if credibility is at all important to you, you will make sure that all the DoNoHarm's claims to that effect are removed from the site.
Subscribe to:
Comments (Atom)